Trehalose is a natural saccharide that is abundant in nature around us.
It has been confirmed that trehalose is contained in most edible fungi and is an important part of reconstituting dried shiitake mushrooms. That’s why it is also referred to as mushroom sugar. Because trehalose is contained in marine plants such as ‘hijiki’ seaweed, it is called seaweed sugar in China.
In fact, trehalose is found in dry yeast, accounting for 12% by dry weight of baker’s yeast. For this reason, dry yeast can be preserved in a dried state without losing fermentative activity. Fermented products including beer and wine contain trehalose. As you can see, we take in trehalose in our daily lives without thinking about it.
that Trehalose is Safe for Human Consumption.
Nagase Viita Co., Ltd., (”Nagase Viita”) manufactures a trehalose product from starch using enzymatic technology and sells it in many countries under the tradename of TREHA®. Essentially all international and national food regulatory authorities have evaluated the safety of trehalose and approved it for use.
As the result of a thorough safety evaluation of trehalose by the Joint WHO/FOA Expert Committee on Food Additive (JECFA), it was concluded that an ‘ADI not specified’ was appropriate for TREHA®, This determination is given to food substances of very low or no toxicity for human consumption as a miscellaneous additive1. In the U.S., Hayashibara (Nagase Viita at present) submitted a GRAS (Generally Recognized As Safe) Notice for trehalose using their enzymatic manufacturing technology (GRN No. 000045) to the U.S. Food and Drug Administration (FDA). The FDA subsequently issued a “No questions” letter for the product. In EU, the Scientific Committee on Food (SCF) evaluated trehalose manufactured using the enzymatic technology, declared it safe, and authorized it to be marketed as a novel food/novel food ingredient within the European Community.




Enzymatic Technology for Food Use in Major Countries
Trehalose manufactured using enzymatic technology is also approved in Australia/NZ (FSANZ), Brazil (ANVISA), Canada (Health Canada), China (MOH), India (FSSAI), and is allowed for use in food products in over 60 countries as of 2021.
Please contact us for information on the regulatory status for any countries of interest.
Trehalose consists of 2 glucose molecules bound by an α,α-1-1 bond. When orally consumed, trehalose is digested by a specific enzyme located in small intestine, trehalase, into glucose, which is absorbed into the body. A large intake of trehalose at one time may induce laxation. The transitory laxative threshold of trehalose was reported to be 0.65 g/kg-body weight, or 39 g in a 60-kg person2. This can occur at lower consumption amounts in a very small percentage of the population having a trehalase deficiency. This is a well-known physiological response similar to lactose intolerance in persons deficient in lactase and/or to intolerance caused by a large intake at one time of non-digestible/non-absorbable saccharides such as sugar alcohols (e.g., sorbitol, maltitol).
For further information on this subject please see the following URL of National Institutes of Health, U.S. Department of Health and Human Services.
Trehalase deficiency | Genetic and Rare Diseases Information Center (GARD)
When Nagase Viita applied for approval of trehalose to international and national authorities, Nagase Viita performed a literature search and reported on the portion of various populations with low trehalase activity. These studies provided a range of low trehalase activity from 0% to a maximum of 2%. The only exception is in a specific closely related genetic population, native Greenlanders (Inuits). In this population about 8% had low trehalase enzyme values; however, they are also known to have approximately 60% lactase deficiency, and even sucrase deficiency which is almost unknown in any other ethnic group3. Please see Table 15 in the U.S. GRAS Notice of trehalose (GRN 00045) which shows the portion of people with trehalase deficiency in the U.S. and EU countries4. JECFA evaluated the same trehalose information concerning people with trehalase deficiency1.
Oku T., Okazaki M. Transitory Laxative Threshold of Trehalose and Lactulose in Healthy Women. Journal of Nutritional Science and Vitaminology. 1998;44:787-798.
Gudman-Høyer E., Fenger H.J., Skovbjerg H., Kern-Hansen P., Madsen P.R. Trehalase deficiency in Greenland. Scandinavian Journal of Gastroenterology. 1988;23:775-778.


PHD,RD,FACSM,CSSD


During the 23rd Trehalose Symposium held in Tokyo on November 7, 2019, Professor Mark Wilcox of the University of Leeds (UK), gave a lecture titled “Trehalose is not associated with the emergence and spread of epidemic Clostridium difficile strains” and explained the contents of his co-authored paper, “Clostridium difficile trehalose metabolism variants are common and not associated with adverse patient outcomes when variably present in the same lineage.”
<DOI:https://doi.org/10.1016/j.ebiom.2019.04.038>
Trehalose is a naturally occurring disaccharide that is found in foods such as mushrooms, shellfish, seaweed (algae) and products made from brewer’s yeast (e.g., beer and breads). Because of its stability and functional properties as well as recent innovations in manufacturing, trehalose has become pervasive in sweet and savory foods, e.g., baked goods, frozen desserts, jams and jellies, rice bowls, salads, meat and fish dishes. Paralleling the increased production and widespread utilization of trehalose has been an increase in the incidence of hospital infections with an antibiotic resistant strain of intestinal bacteria—Clostridium difficile (C. difficile). Researchers from Baylor College of Medicine and the University of Oregon have tried to link these two seemingly arbitrary occurrences together in a series of epidemiological and microbiological studies, which has led to media reports and cautionary advice to avoid dietary trehalose (Collins et al. 2018 & Collins et al. 2019). But are these warnings warranted?
Trehalose is a disaccharide comprised of two glucose molecules bonded together. What differentiates trehalose from the more common disaccharide maltose (also consisting of two glucose molecules bonded together) is the bond that connects the glucose molecules. That is, trehalose contains “an α,α-1,1 glycosidic bond,” while maltose contains “an α-1,4 glycosidic bond.” Additional factors that differentiate the two glucose containing disaccharides include the fact that maltose is a reducing sugar, whereas trehalose is not, and trehalose is significantly more “stable” than maltose under high temperatures and acidic pH (Richards et al.2002).
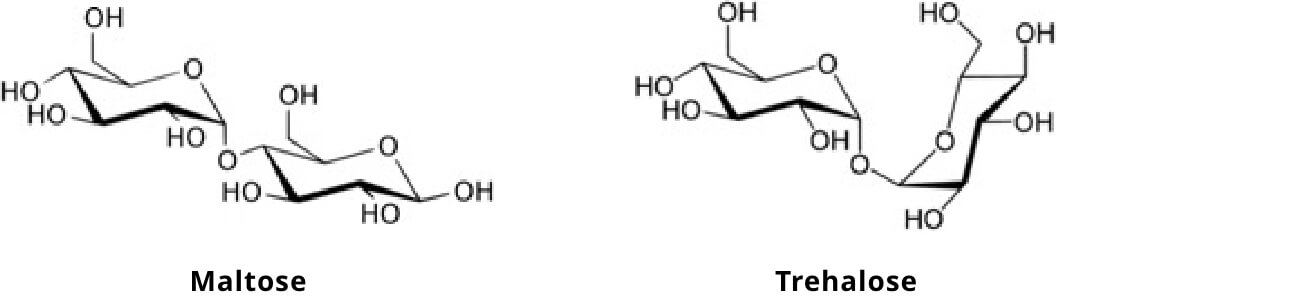
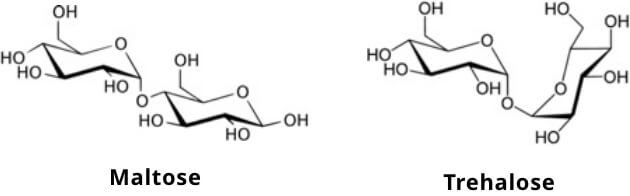
The digestion and absorption of trehalose in humans is similar to that of other disaccharides. Trehalase, an enzyme produced and secreted from the small intestine brush border, cleaves the glycosidic bond, releasing two molecules of glucose which are subsequently actively absorbed by the intestinal mucosal cell via sodium glucose transporter 1 (SGLT 1) (Elbein 1974 & Richards et al. 2002).
Trehalose occurs naturally in small amounts in a variety of foods most notably mushrooms, honey, lobster, shrimp, certain seaweeds (algae), wine, beer, bread and other foods produced by using baker’s or brewer’s yeast (Richards et al. 2002). It can also be manufactured from a variety of plant-based starches using enzymatic processing procedures (Maruta et al. 1995). Because it is heat-stable, hydrophilic and resistant to acidic hydrolysis, trehalose has been used a texturizer, stabilizer and occasionally a sweetener in dried foods, frozen foods (e.g., ice cream), nutrition bars, fruit fillings and jams, instant noodles and rice, baked goods and fruit juices (Richards et al. 2002). Trehalose gained “generally recognized as safe” (GRAS) status by the U.S. Food and Drug Administration in 2000 and was approved for use in foods in Europe in 2001 (Richards et al. 2002).
In January of 2018, a paper was published in the journal Nature linking trehalose to a particularly “virulent” (i.e., infectious) strain of C. difficile (Collins et al. 2018). Specifically, the paper summarized a series of in vitro and animal experiments showing that while the virulent strain can thrive on a number of different carbon sources (e.g., simple sugars, sugar alcohols, amino acids and some small proteins), it is able to grow robustly on low doses of trehalose. According to the authors of the Nature paper, this finding was the “smoking gun”—the conclusive evidence for the microbiological role of trehalose in the growth and proliferation of the infectious strain of C. difficile (Abbasi 2018). This data, combined with observations that increased production and utilization of trehalose in the food supply coincided with the rise of infections caused by the virulent strains of C. difficile, led to speculation that trehalose caused the epidemic emergence of the virulent strain of C. difficile (Collins et al. 2019).
The short answer is “no.”
The long answer requires a slightly more in-depth evaluation of the existing research on trehalose, beginning with the Nature paper.
The Nature paper is not a single study, but rather a compilation of summaries of individual experiments each conducted by one or more of the authors listed on the paper. Several experiments described in the paper simply examined the genetic variants of the different strains of C. difficile, including the most virulent strains, to characterize the microbiology and physiology. Another experiment tested several carbon sources (i.e., various carbohydrates and amino acids) to determine if they supported the growth of a particular virulent strain of C. difficile in vitro (that is, in a petri dish). Interestingly, the findings indicated that several carbohydrates and amino acids (not just trehalose) caused a 1.5-fold increase in the virulent strains. Nonetheless, the researchers chose only to focus on trehalose, allegedly because unlike some of the other carbon sources studied, trehalose supported growth even at relatively low concentrations. The experiment that probably garnered the most media attention involved feeding mice either trehalose or water. The results showed that the trehalose-fed mice had an increase in the growth of the resistant strains of C. difficile. These results are not surprising. Of course, we would expect to see greater growth of C. difficile on a carbohydrate source compared with water. A better, more equitable comparison would have been another sugar (or more than one sugar), particularly one that has been shown in previous experiments to stimulate the growth of C. difficile.
One other final experiment in the Nature paper that is worth noting examined the metabolism of trehalose in the intestines of three human subjects consuming their “normal diets” (which strangely were never described in the paper). The results of this final experiment showed that in two of the three subjects, there was an increased expression of the “treA gene,” which is required to metabolize trehalose. No statistics were run on this “sample” likely because it was too small and there really wasn’t anything to compare or calculate. In addition, the study did not show that eating a “normal diet” containing trehalose increased levels of C. difficile bacteria in the gut, nor that it caused symptoms; only that it increased the expression of a gene that metabolizes trehalose.
Thus, the data reported in this paper does not show that trehalose causes the growth and proliferation of resistant strains of C. difficile in humans. It does show that the resistant strains of C. difficile can grow in vitro on low doses of trehalose, but they metabolize and flourish on several other carbon sources as well (and, as noted, none were directly compared to trehalose). Moreover, growth in vitro in a laboratory setting does not necessarily generalize to a human gut. The “feeding” experiment was conducted in a mouse model (which may or may not translate to a human model) and did not include a carbohydrate comparison. Finally, the only in vivo experiment described in the Nature paper included just three subjects and measured changes in the expression of a gene that metabolizes trehalose (not development or proliferation of C. difficile).
More recent research not only contradicts findings from the Nature paper, but refutes a causal connection between trehalose consumption and C. difficile infections.
Eyre and colleagues (Eyre et al. 2019) examined the potential for trehalose metabolism variants to confer a select advantage for the virulent strains of C. difficile by evaluating how common these variants are within the total genetic diversity of clinical C. difficile using previously sequenced isolates. In addition, the researchers compared the effects of trehalose, glucose or saline supplementation on C. difficile metabolism using a validated and clinically reflective gut model of C difficile. The results indicated that trehalose metabolism/utilization variants are quite common among the population and supplementation of trehalose did not result in increased levels of C. difficile or its spores compared with glucose or saline supplementation. Quite the contrary, supplementation with trehalose actually reduced toxin detection to undetectable levels.
In a recently published case-controlled study (Saund et al. 2020) evaluated the potential contribution of trehalose consumption to clinical outcomes in 1144 hospitalized patients with C. difficile. After controlling for all clinical factors independently associated with risk for severe infection outcome, the authors found no significant association between the presence of trehalose utilization variants in infectious C. difficile strains and the development of severe infection outcomes.
Notably in both studies, trehalose metabolism/utilization variants were more widespread than previously thought, suggesting that the ability to utilize low concentrations of trehalose was acquired prior to the recent increase in trehalose production and utilization (Eyre et al. 2019 & Saund et al. 2020).
While the oft-cited association between trehalose production/utilization and C. difficile proliferation may seem compelling, other epidemiological observations suggest that factors besides trehalose consumption may be contributing to the increase in virulent strains of C. difficile (Buckley et al. 2021). For example, in Canada, outbreaks of the virulent strains of C. difficile occurred some three years before the approval and expanded use trehalose. In addition, data from the U.S. Center for Disease Control (CDC) indicates that at least one of the virulent strains of C. difficile is actually declining, despite the fact that no active measures have been taken to reduce trehalose production or consumption (Abasi 2018). Finally, and probably most convincingly, research evaluating the extent to which imports of synthetic trehalose altered total dietary trehalose intake during the rise of the virulent strains of C. difficile in Europe, the U.S. and Canada and showed that the increase in trehalose imports postdated the start of C. difficile epidemics in these countries by at least five years (Eyre et al. 2019).
Trehalose is a naturally occurring disaccharide that has been consumed by humans for centuries and used in food production for decades—long before the epidemic rise of virulent strains of C. difficile. While in vitro data shows that virulent strains of C. difficile proliferate when exposed to low levels of trehalose, in vivo data in humans using appropriate comparisons and controls is generally lacking and the few existing studies have failed to confirm these results. Finally, the majority of existing epidemiological research does not support a valid association between trehalose production/usage and the emergence of resistant strains of C. difficile. Thus, the preponderance of evidence indicates eating foods containing trehalose is safe and will not significantly increase the risk of developing resistant strains of C. difficile (Buckely et al. 2021).
In a recent publication covered by the media, Collins et al. reported that a rise in trehalose consumption could explain increased rates of mortality from Clostridium difficile infections. Scientists were skeptical of the claims, and sought to clarify this important scientific question: is trehalose the cause of increasing C. diff virulence? A new publication led by Professor Mark Wilcox (MD) from the University of Leeds/Leeds Teaching Hospitals aimed to fill in the gaps of the previous research paper, and to test their claims under more robust settings. Prof. Wilcox’s study in fact showed that trehalose is not responsible for increased virulent strains of C. diff. Prof. Wilcox presented his findings at the 2019 Trehalose Symposium in Tokyo, Japan to a group of industry leaders and researchers interested in and working with trehalose, explaining crucial gaps in the Collins et al. paper, such as inaccurate conclusions about the correlation between trehalose consumption and epidemic strains of C. diff, whether bacteria with variant metabolism of trehalose are only commonly found in virulent strains, and if in fact, trehalose impacts C. diff growth or toxin production in a human gut model.
- Trehalose is a naturally occurring disaccharide that is readily digested in our guts. A very small portion of the population may have impaired digestion of trehalose, but this is very uncommon.
- Many strains of C. diff carry the genetic mutation to metabolize trehalose, not just epidemic strains as claimed by Collins et al.
- A strain of C. diff that has the ability to metabolize trehalose does not result in more deaths than a strain of C. diff that cannot metabolize trehalose.
- There is no correlation between the importation of trehalose and the rate of epidemic strains of C. diff.
- Trehalose does not stimulate C. diff growth in an infected human gut model. In fact, supplementation with trehalose in an infected human model led to a reduced detection of C. diff toxin to undetectable levels.
Trehalose is a naturally occurring disaccharide made up of two glucose molecules. It occurs naturally in foods such as mushrooms, honey, shrimp, yeast, and soybeans. Because of its unique culinary properties, it is also a highly desirable disaccharide used in a variety of food service applications to enhance freshness and flavor.
Until the discovery of mass production in Japan, it was very difficult to produce trehalose. With the ability to produce this desirable sugar, it is now exported to a variety of different countries and used in many culinary applications.
In humans, trehalose is a highly digestible disaccharide – meaning it is broken down to 2 glucose molecules and absorbed into the blood stream, not reaching the colon. A very small portion of the population (~8% of Greenlandic individuals) may carry a genetic abnormality that prevents digestion of trehalose and results in symptoms similar to lactose intolerance.
Clostridium difficile causes a type of hospital-acquired infection that is characterized by diarrhea and inflammation of the colon. This can pose serious consequences for patients, including prolonged hospitalization and increased mortality. C. diff infections are on the rise in some countries - both the number and severity of cases and can often be difficult to manage.
The change in the epidemiology of C. diff infection is in part due to emergence of certain strains such as ribotype-027 and 078, which have been associated with increases in morbidity and mortality. With the rise in C. diff virulence, researchers are working quickly to understand the cause and find solutions to this rapidly evolving disease.
Collins et al. research claims that trehalose imports have played a significant role in the emergence of epidemic strains of C. diff. However, upon careful analysis, several concerning gaps in the research are identified by Wilcox et al. that actually disprove this theory.
We are going to outline limitations of this research and discuss why consumers need not fear trehalose, a commonly consumed disaccharide, both natural and added, in our diet.
Certain strains of C. diff carry a genetic mutation that allow them to, as Prof. Wilcox describes, “more effectively metabolize trehalose; breaking trehalose into glucose, and using the glucose as a growth food substance. And that if it can do that, the C. diff strain will be able to outcompete potentially, other C. diff strains and maybe other bacteria if we find C. diff in the human gut.”
On this hypothesis, Prof. Wilcox and his team set out to determine if this mutation was common only to the epidemic strains, conferring a selective advantage as Collins et al. claimed, by reviewing the genetic code of over 10,000 strains of C. diff for the trehalose metabolizing variant/mutation.
Describing his findings, Prof. Wilcox states: “we see that actually the trehalose mutations, the gene mutations, are very common. They are spread through lots of different types of C. diff, not just epidemic types. And, because of the way they are distributed, we conclude that these mutations are ancient; very old mutations that happened hundreds or thousands of years ago. So the original claims that were made by Collins et al. about these mutations being specific to epidemic C. diff strains - 027, 078, 017 - are not true.”
This is important because it calls to question the claim that these genetic mutations are the driving force in the C. diff ribotype-027, 078, 017 epidemics, which leads up to the important question - does the ability of certain strains of C. diff to metabolize trehalose make a difference to patient outcomes?
| Factor | Alive, n (%)/ median (IQR) | Died, n (%) / median (IQR) | Univariable OR (95% CI) | p value | Multivariable OR (95% CI) | p value |
|---|---|---|---|---|---|---|
| n | 181 | 27 | ||||
| Age, years OR per 10-year increase | 73 (59-82) | 84 (69-88) | 1.42 (1.06-1.90) | 0.02 | 1.45 (1.08-1.34) | 0.01 |
| Sex | ||||||
| Female | 110 (88%) | 15 (12%) | ||||
| Male | 71 (86%) | 12 (14%) | 1.23 (0.55-2.80) | 0.61 | 1.27 (0.55-2.95) | 0.58 |
| ST | ||||||
| ST44 | 103 (88%) | 14 (12%) | ||||
| ST10 | 78 (86%) | 13 (14%) | 1.23 (0.55-2.76) | 0.62 | 0.55 (0.15-2.00) | 0.36 |
| Four-gene cluster | ||||||
| Absent | 93 (85%) | 17 (15%) | ||||
| Present | 88 (90%) | 10 (10%) | 0.62 (0.27-1.43) | 0.26 | 0.36 (0.09-1.34) | 0.13 |
To see whether this genetic mutation is a causal factor in increased mortality, Wilcox et al. utilized C. diff ribotype-015; some of the strains in this ribotype carry the metabolic mutation for trehalose metabolism and some do not. This is unlike ribotype-027 strains, which all appear to have the mutation. This allowed researchers to test if those infected with a trehalose metabolizing strain of C. diff had worse outcomes than those infected with a non-metabolizing strain. Prof. Wilcox and his team found that “there is no association between possession of this trehalose mutant gene metabolism, four-gene cluster, and death, likelihood of death, in humans infected with these strains. The mutation does not appear to be responsible for increased risk of death in human infection.”
Many strains of C. diff carry the genetic mutation to metabolize trehalose, not just epidemic strains. A strain of C. diff that has the ability to metabolize trehalose does not result in more deaths than a strain of C. diff that cannot metabolize trehalose.
Interestingly, Collins et al. suggests correlation between the increasing importation of synthetic trehalose with a rise in C. diff infection, claiming that “the widespread adoption and use of trehalose in the diet coincides with the emergence of both ribotype-027 and 078 outbreaks”. It is important to know that correlation does not equal causation, and in fact, when you look closer at trehalose importation and virulent C. diff out breaks, they do not correlate at all.
Collins et al. estimate that trehalose intake from naturally occurring sources is approximately 100 grams per capita annually. Between 2000 and 2006, imported levels of trehalose were <1 gram per capita per year in the USA and England, not substantially increasing trehalose intake.
He points out that while both strains ribotype-027 and 078 existed prior to 2001, that epidemic outbreaks did not occur until 2003, and include major outbreaks in both Quebec, Canada, and Pittsburgh, USA. He goes on to suggest a correlation of these major outbreaks with trehalose imports and use.
However, synthetic trehalose was not approved for use in Canada until 2005, and imports to the US did not start increasing until 2007, showing, as Prof. Wilcox described, that it’s “not possible to claim that importation of trehalose was associated with the epidemic of ribotype-027.” Trehalose imports postdate the start of the 2003 epidemic by 3 to 4 years and amounts of added trehalose per capita are extremely low compared with naturally occurring intakes of trehalose; this shows that there is no correlation between trehalose imports and the emergence of ribotype-027 and 078 outbreaks.
There is no correlation with importation of trehalose and correlation of epidemic strains of C. diff. Increasing intake of added trehalose postdate the initial epidemics in question.
Lastly, Prof. Wilcox and his team set out to describe how C. diff would grow in a human gut model (a triple phase chemostat gut model) when trehalose is administered (as a potential food for the bacteria) in comparison to controls. Using a model that has been proven to simulate the human intestine and gut microbiota, the team infected several of these with C. diff and administered different substrates, including trehalose, glucose, or saline, to test this theory.
Interestingly, in a human model, trehalose suppressed the production of toxins, when compared with both the glucose and saline model, demonstrating that trehalose consumption is not associated with increased C. diff virulence in a human model.
As Prof. Wilcox explains:
“If trehalose is meant to be a stimulant, an inducer of infection in humans, as claimed in the Nature publication, why do we see no toxin production at all? Our conclusion again is that the data in the Nature publication are not accurate and the claims are not true about trehalose and virulent C. diff. The earlier experiments were done in mice, whereas ours were carried out in a model that simulates human C. diff infection. Notably, we have known for many years that C. diff infection, whether it occurs or not, is specific to the animal type.”
Collins et al. state “the ability to metabolize trehalose at lower concentrations confers a competitive growth advantage in the presence of a complex intestinal community”, however this was demonstrated in an animal model, which was not repeatable in a proven human model. In fact, supplementation with trehalose led to a reduced detection of C. diff toxin to undetectable levels, meaning that trehalose does not appear stimulate C. diff growth in humans.
Trehalose does not stimulate C. diff growth in an infected human model.
In fact, supplementation with trehalose in an infected human model suppressed production of C. diff toxin to undetectable levels.
Through Prof. Wilcox’s research and presentation at the Trehalose Symposium, it is clear that trehalose is not associated with the rise in epidemic strains of C. diff. Not only was Prof. Wilcox able to demonstrate that strains with a genetic mutation to metabolize trehalose have no impact on morbidity and mortality of C. diff, he was also able to demonstrate that trehalose had no bearing in a human model of C. diff infection.
In summary, we can say with confidence that there is no relationship between the importation and consumption of trehalose in humans and the emergence of epidemic strains of C. diff.
We are grateful for Professor Mark H. Wilcox for the support and proofreading.
We are grateful for Andrea Hardy, RD for preparation and suggestions.
The contents are prepared with the support of Andrea Hardy, Registered Dietitian in Calgary, Canada, in response to the following articles:
• Nature 553, 285-286 (2018) DOI: https://10.1038/d41586-017-08775-4
• EBioMedicine 43 (2019) 347–355 DOI: https://doi.org/10.1016/j.ebiom.2019.04.038
with the information based on the speech of Prof. Mark H. Wilcox at The 23rd Trehalose Symposium held in Tokyo on 7th November, 2019.




